iGEM REPORT: Investigation of glutathione reductase and 25-hydroxylase for clearing aggregated proteins in an experimental fish eye cataract model
Note: This iGEM Report was submitted to the PLOS iGEM Realtime Peer Review Jamboree, and has not undergone formal peer review by any of the PLOS journals. We welcome your comments on this work.
Investigation of glutathione reductase and 25-hydroxylase for clearing aggregated proteins in an experimental fish eye cataract model
Fiona Tsai (1), Huiru M Huang (1), Chang Sun Lee (1), Jason Hsu (1), Megan Yang (1), Avery Wang (1), Leon Yim (1), Alvin E Wang* (1), Chih Heng Lai (1), Angela Y Lu (1), Jacqueline Chein (1), Hellen Jang (1), Austin Lai (1), Apollo Lo (1), Justin Chang (1), Moksha Shah (1), Kevin Liao (1), Justin J Yang (1), Catherine Yeh (1), Yvonne Wei (1), Jubi Y Lin (1), Andrew Chen (1), Dennis Jang (1), Zach Verne (1), Stephanie Cheng (1), Sean Tsao (1), Teresa Chiang (1), Jude C Clapper (1)
- Taipei American School, Taipei City, Taiwan.
*Corresponding author: [email protected]
Author Contributions
Conceptualization: FT HH MS CSL AW AEW MY DJ LY CHL AL AYL JC HJ AL JC KL JY CY YW JL AC ZV SC
Methodology: AW FT HH AEW
Software: AW
Investigation: FT HH MS CSL AW AEW MY DJ LY AL AYL JC HJ AL JC KL JY CY YW JL AC ZV SC
Resources: JCC TC
Writing – Original Draft: FT HH MS CSL AW MY DJ AEW
Writing – Review & Editing: FT HH MS CSL AW MY DJ AEW TC JCC
Visualization: JY AW
Supervision: ST TC JCC
Project Administration: JCC
Funding Acquisition: JCC
Abstract
Cataracts, areas in the lens which become cloudy, contribute to half of the world’s blindness. Most cataracts are age-related and arise when crystallin proteins in the lens become oxidized and aggregate over time. Surgery is currently the only recognized treatment to effectively cure cataracts, but this method is expensive and invasive. In this paper we investigate the ability of the proteins glutathione reductase and 25-hydroxylase to clear aggregated proteins in an experimental fish lens cataract model. In the eye, a natural antioxidant glutathione—produced by the enzyme glutathione reductase—exists to combat oxidation, but glutathione levels decrease with age. Studies have also suggested that another enzyme—25-hydroxylase—can convert cholesterol in the eye into 25-hydroxycholesterol, which reverses crystallin aggregation. We simulated cataract formation in fish lens proteins using hydrogen peroxide, and in this model we find that both glutathione and 25-hydroxycholesterol decrease the cloudiness of fish lens solutions. We also created plasmid constructs to express recombinant glutathione reductase and 25-hydroxylase. In addition, we investigate the ability of chitosan nanoparticles to both encapsulate and release proteins as a potential delivery option. Although our current work remains experimental, with further research we envision that delivery of recombinant proteins to the eye may have potential to treat or prevent cataract formation.
Financial Disclosure
This work was funded by the Taipei American School. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing Interests
The authors have declared that no competing interests exist.
Data Availability
Yes – all data are fully available without restriction. Sequences for the plasmids used in this study are available through the Registry of Standard Biological Parts. Raw data are included in the Supplementary Information.
Introduction
Cataracts are the leading cause of blindness today, affecting 20 million people worldwide (1). Half of Americans above 80 years old are affected by cataracts. The National Eye Institute projects that in 30 years, the number of cataract patients will increase to 50 million (2). However, the current standard treatment for cataracts is invasive and requires trained surgeons with professional equipment. These requirements add to the cost of surgery, which averages about $3,500 per eye in the US without insurance (3).
Cataracts can be caused by many factors, including radiation and diabetes, but the underlying cause is oxidative damage. Oxidative damage occurs when reactive oxygen species, or unstable chemicals containing oxygen, react with DNA, lipids, or proteins, disrupting cellular functions (4). In the lens, crystallin proteins can be oxidized by hydrogen peroxide (H₂O₂), a reactive molecule produced during aerobic respiration (5). H₂O₂ reacts with amino acid residues and alters protein shape. Damaged crystallin proteins thus aggregate and form clumps in the lens (4). In the eye, a natural antioxidant called glutathione (GSH) exists, which converts H₂O₂ into water (6). With age, however, GSH levels decrease (as GSH itself becomes oxidized to form GSSG, which has no antioxidant effects). With decreased GSH, H₂O₂ accumulates in the lens, causes oxidative damage, and leads to the development of cataracts. In this study we engineered a construct that codes for glutathione reductase (GSR), an enzyme that recycles GSSG back into GSH.
If cataracts have already formed, however, then treatment is needed to restore lens transparency. It has been previously shown in mice that a molecule called 25-hydroxycholesterol (25HC) can reverse the aggregation of crystallin protein clumps to improve lens transparency (7). To produce 25HC, we engineered a construct expressing cholesterol 25-hydroxylase (CH25H) an enzyme which converts cholesterol into 25HC.
We mimicked the formation of cataracts in fish lens proteins, and used this model to test the prevention and treatment effects of GSH and 25HC, respectively. Finally, we envision delivering the proteins GSR and CH25H into the lens using nanoparticles (8), and synthesized chitosan nanoparticles to test this concept. Mathematical modeling was also used to simulate drug release from nanoparticles. Our long-term aim, through further research, is to develop a safe and cost-effective cataract treatment that may serve as an alternative to surgery.
Materials and Methods
Setting up a cataract model
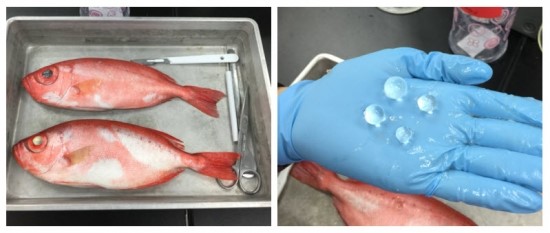
We purchased dead Priacanthus macracanthus from a fish market and extracted soluble proteins from the lens. We removed the lens cortex and obtained the nucleus because the nucleus contains older cells and is more prone to cataract formation (9). In order to solubilize lens proteins, the lens nucleus (in Tris buffer) was placed in a shaking incubator overnight (10). After centrifugation, the supernatant containing soluble lens proteins was left either untreated or treated with 1mM and 10mM hydrogen peroxide (H₂O₂).
Measuring absorbance
To quantify the severity of cataracts in our model, we used a spectrophotometer to measure absorbance values of the lens solution. As lens proteins become oxidized, absorbance should increase because as proteins aggregate, they fall out of solution, and will scatter incoming light. To find a wavelength of light for data collection, we compared the absorbance of normal lens solution (negative control), 10mM H₂O₂ lens solution, and heat-denatured lens solution (positive control). We observed an absorbance peak at 397.5 nm for both H₂O₂-treated and heat-denatured solutions, and chose to collect all future absorbance values at that wavelength. Using SDS-PAGE, we confirmed that the 10mM H₂O₂ lens solution exhibited protein aggregation.
Prevention of cataracts with glutathione
We purchased GSH from Sigma Aldrich. Three samples of lens solution (3 mL each) were incubated in 10 mM H₂O₂ and 8 mg GSH. In the negative control, only H₂O₂ was added. We measured the absorbance of the solutions at 0, 24, and 48 hours.
Treatment of cataracts with 25HC
We purchased commercial pet eye drops (OcluVet), which is advertised to reduce cataracts, and 25HC from Sigma Aldrich. OcluVet lists L-carnosine, N-acetyl L-carnosine, L-taurine, glutathione, and cysteine ascorbate as the active ingredients (11). We compared the effects of 25HC and Ocluvet in our fish lens model. First, lens solutions were incubated with 10 mM H₂O₂ for 24 hours to induce cataract formation. 25HC (dissolved in ethanol) was added to the lens solutions to reach concentrations of 20 uM or 50 uM. In another sample, 3 drops of OcluVet, the recommended daily dosage, was added for comparison. We measured the absorbance of the samples at 0, 24, and 48 hours.
Construct design
GSR construct
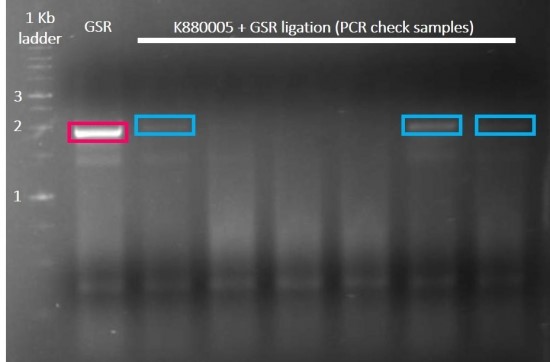
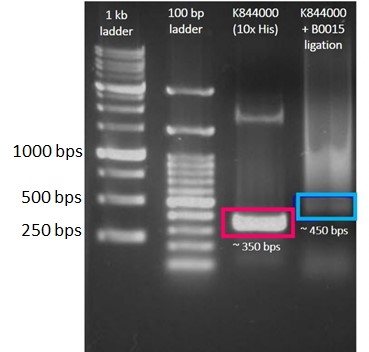
We designed a construct including a strong promoter, strong ribosome binding site, GSR (mutated to remove internal restriction sites), 10x histidine-tag, and a double terminator (Figure 1, top). We acquired a strong promoter and strong ribosome binding site part (BBa_K880005) from the iGEM distribution kit to maximize protein production. We ordered the cDNA of GSR from Origene and used the 10x histidine-tag part from the distribution kit (BBa_K844000). A double terminator (BBa_B0015) was added at the end to stop transcription. The purchased GSR cDNA had two internal PstI and three EcoRI cutting sites. After making silent mutations to the sequence, we sent the cDNA to Mission Biotech for mutagenesis to remove these cutting sites. Once we had the correct sequence of GSR (with 5 point mutations), we designed primers to add the BioBrick prefix and suffix in order to clone GSR into a BioBrick backbone. The primer designs were sent to Tri-I Biotech for oligo synthesis, and polymerase chain reaction (PCR) was set up. GSR was inserted behind BBa_K880005 (strong promoter + strong ribosome binding site), then inserted before the 10x his-tag and double terminator. Sequencing results from Tri-I Biotech show that our final construct was correct.
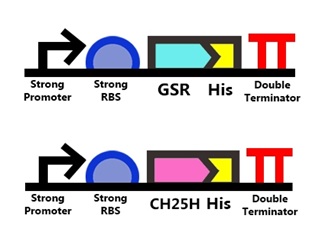
CH25H Construct
The CH25H construct contains similar components as the GSR construct, except the GSR gene is replaced by CH25H (Figure 1, bottom). CH25H was mutated (by Mission Biotech) to remove internal cutting sites and put into a Biobrick backbone to make a new basic part. The sequence of the final construct was sent to Integrated DNA Technologies for synthesis and cloned into a Biobrick backbone.
Chitosan nanoparticle synthesis
We dissolved 3 mg/mL low molecular weight chitosan in 1% (by volume) glacial acetic acid. We adjusted the pH of the chitosan solution to 5.5 by adding 1M sodium hydroxide, then dissolved 4 mg of desired protein for each millimeter of chitosan solution. We then added proteins including bovine serum albumin (BSA), green fluorescent protein (GFP), red fluorescent protein (RFP), and green pigment protein (pGRN). 1 mg/mL of sodium triphosphate was dissolved in water. We stirred the chitosan solution at 600 rpm while adding an equal volume of sodium triphosphate solution dropwise. To isolate nanoparticles, the suspension was centrifuged at 17000 xg for 40 minutes at 4 degrees Celsius. We imaged the pellet using atomic force microscopy to confirm nanoparticle formation.
Protein encapsulation
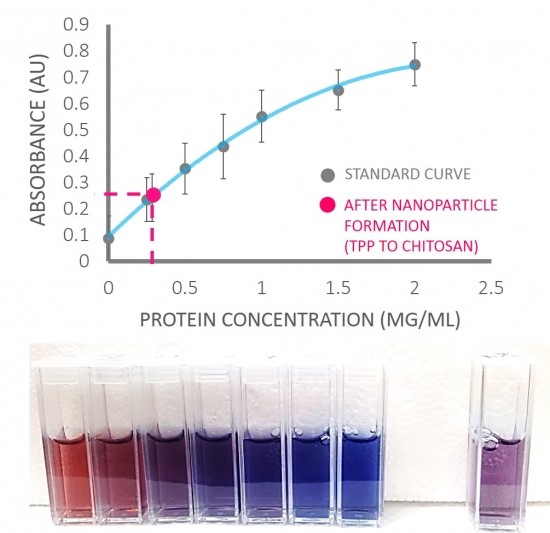
We synthesized chitosan nanoparticles with and without BSA, and collected the supernatants after centrifugation. We added known amounts of BSA to the blank supernatant to create standard solutions. Using a Bradford assay, we then measured the protein concentration in the supernatant of BSA-containing nanoparticles to measure the amount of proteins that failed to be encapsulated. We assume that the amount of encapsulated protein is the initial amount of protein subtracted by protein remaining in the supernatant. The BSA encapsulation efficiency was calculated using the equation below:
BSA encapsulation efficiency =
(initial BSA concentration – BSA concentration in supernatant)/(initial BSA concentration) * 100
Sample calculation:
Initial BSA concentration added: 2 mg/mL
BSA concentration in the supernatant: 0.49 mg/mL
Encapsulation efficiency= (2 – 0.49)/(2) * 100 = 75.5%
Protein release measurements
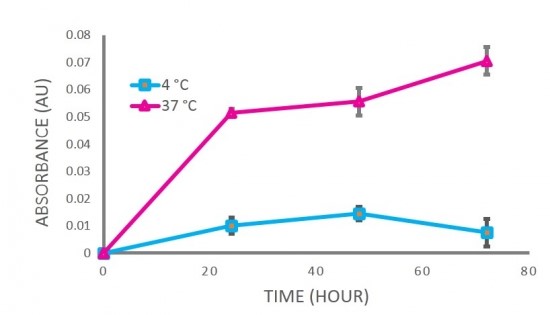
We resuspended nanoparticles in phosphate buffered saline (12), and placed samples at different temperatures: 4°C and 37°C. As the nanoparticles degraded, they released proteins into the saline solution. Using a Bradford assay, we measured protein concentration in the saline solution at 0, 24, 48, and 72 hours after incubation.
Long-term nanoparticle model
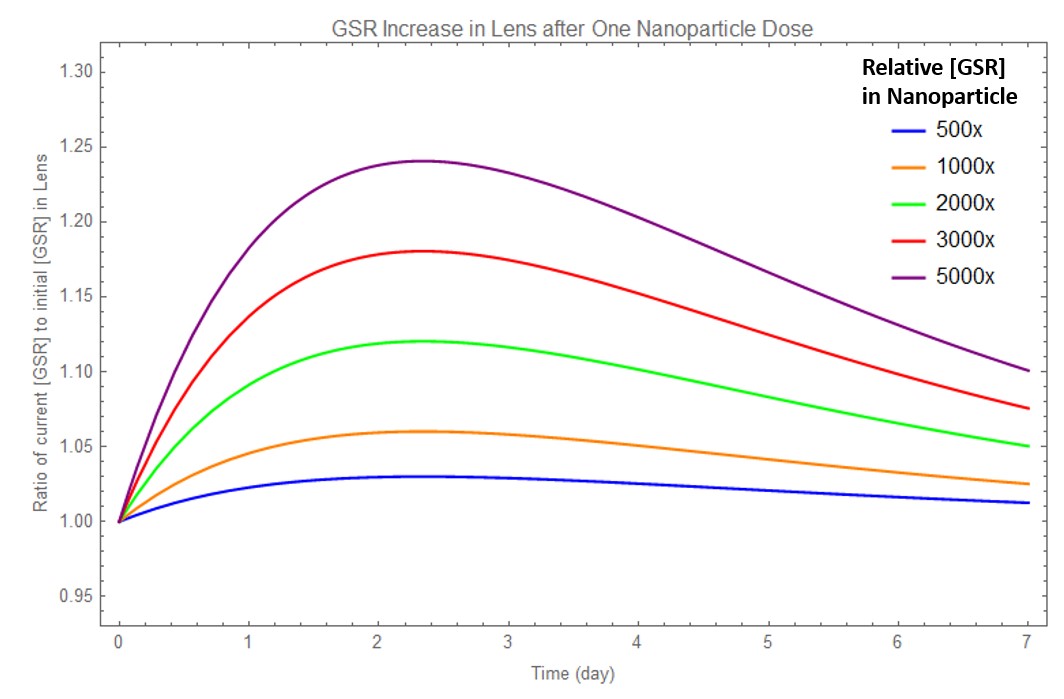
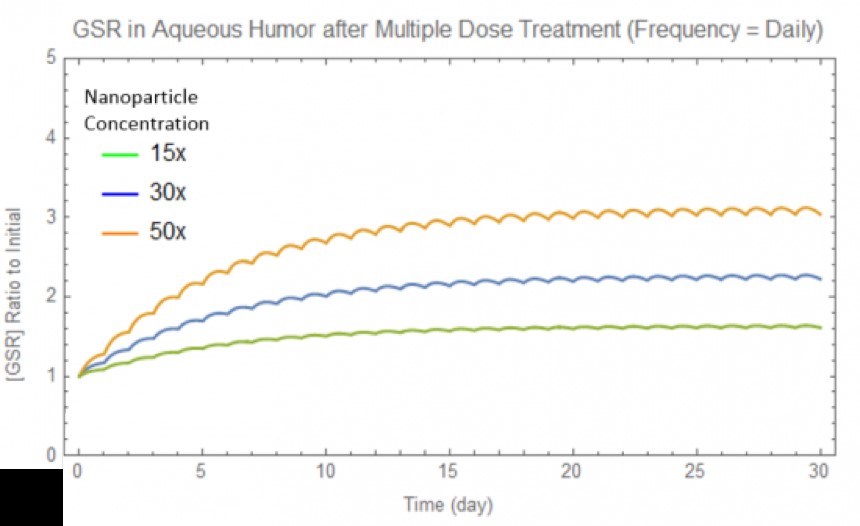
We cannot experimentally test the behavior of our nanoparticle treatment over a long period, so we built a mathematical model to analyze the amount of GSR maintained in the lens over time, as eye drops are regularly applied. First, we created a model of the release of a single dose of nanoparticles using the Noyes-Whitney equation (13). We sum up the contributions of each dose of nanoparticles which correspond to each application of the eye drop, yielding the final model of GSR concentration over time. We can alter variables, such as concentration, eye drop frequency, and nanoparticle radius, to analyze their impacts on GSR concentration in the lens.
Chitosan nanoparticle-embedded contact lenses
We found a method to make chitosan nanoparticle-embedded hydrogel contact lenses (14). Following the protocol, we created a polymer solution containing all the necessary components. We dispersed GFP-nanoparticles in the polymer solution to test if these protein-containing nanoparticles could be embedded in the contact lenses. We then transferred this solution into a 3D-printed mold. After exposure to UV for 40 minutes, we successfully made hydrogel contact lenses.
Results and Discussion
First, we wanted to investigate whether GSH and 25HC could decrease the cloudiness of fish lens solutions that mimic cataract formation. We chose to use fish lenses because fish are commercially available, and their lenses contain similar crystallin proteins (15). We set up the cataract model by extracting soluble proteins from fish lenses (Figure 2), and then oxidizing the proteins with H₂O₂. As lens proteins become oxidized, absorbance should increase because the proteins will aggregate and scatter incoming light. Our results show that increasing concentrations of H₂O₂ lead to higher absorbance, or opacity, of the lens solution (Figure 3). We also ran an SDS-PAGE gel to compare the proteins from untreated and H₂O₂-treated solutions (Figure 4). After treatment with H₂O₂, there was an increase in higher bands, which suggests that H₂O₂ causes proteins to clump and aggregate. Together, the SDS-PAGE and our absorbance data show that our experimental model accurately represents cataract development.
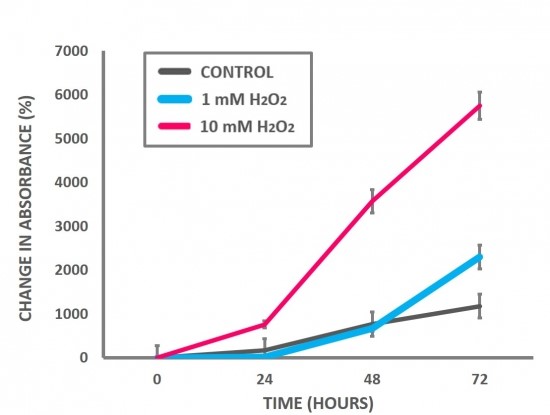

Using the cataract model, we demonstrated that GSH and 25HC can respectively prevent and reduce the clouding of fish lens solutions. GSH-treated solutions showed lower absorbance values compared to the untreated control, indicating less protein aggregation (Figure 5). These results suggest that GSH has a preventative effect on cataract formation. 25HC was shown to reduce the absorbance of lens solution that already reacted with H₂O₂. In addition, a higher concentration of 25HC lowered the absorbance even further (50 μM compared to 20 μM). We compared 25HC to commercial pet eye drops, which did not lower the absorbance, suggesting that 25HC is more effective (Figure 6). Although our results demonstrate that 25HC can reduce the opacity of H₂O₂-treated lens solutions, researchers do not fully understand the mechanism.


Since GSH and 25HC are produced by GSR and CH25H respectively, we designed and constructed plasmids to produce his-tagged GSR and CH25H (Figures 1). Examples of DNA gel electrophoresis after PCR check are included in Figures 7 and 8.
The cornea protects the eye from foreign materials, but it also prevents drugs from reaching the lens (16). Researchers have developed several methods to penetrate the cornea for drug delivery, but many of these techniques are invasive, such as inserting implants (17). One of the most promising and noninvasive methods is using chitosan nanoparticles as drug carriers (18). Researchers have tested the use of chitosan nanoparticles in the eye; their low toxicity to somatic cells makes them safe and they do not affect the anatomy of the eye (19). They can embed in the cornea, and their biodegradability allows a packaged drug to be released continuously into the eye (20).
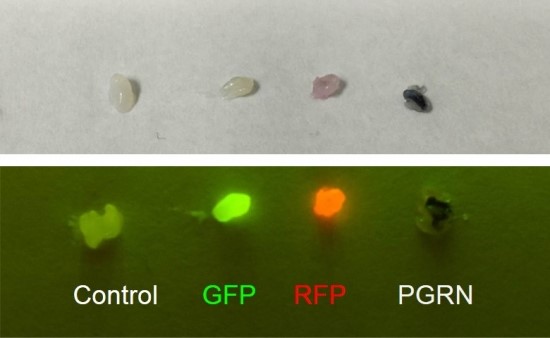
We synthesized and imaged chitosan nanoparticles using scanning electron microscopy (Figure 9) and atomic force microscopy (Figure 10). We find that our nanoparticles range from 100 to 400 nm in diameter. We also show that they can be used to encapsulate proteins (Figure 11). When GFP- and RFP-containing nanoparticles were exposed to blue light, we find that the proteins still fluoresce, which indicate that the proteins remain functional after the encapsulation procedure. We show that our chitosan nanoparticles encapsulate proteins at 72% efficiency (Figure 12), and can subsequently release the protein contents. Proteins were released from nanoparticles (into the surrounding solution) when samples were incubated at 37°C for 72 hours, but minimal protein release was observed at 4°C (Figure 13). This finding suggests that we can store a final functional product (e.g., eye drop) at 4°C without nanoparticle degradation, while the proteins can be released from nanoparticles when the eye drop is applied at body temperature.


Using experimental data from the BSA protein release from chitosan nanoparticles, we used computer modeling to show how GSR concentration in the eye would increase after application of the eyedrop (Figure 14). The GSR concentration approaches a limit after about 3 weeks, and further eye drop use will maintain the GSR concentration. This limit is proportional to the concentration of GSR in the nanoparticles (Figure 15). Additionally, modelling suggests that it is better to apply small concentrations of nanoparticles frequently as opposed to larger concentrations occasionally (Figure 16), as we want to have consistent GSR concentrations within the eye. This modeling data may be valuable to manufacturers and clinics for drug delivery using eye drops in the future. We also considered the application of proteins using nanoparticle-embedded contact lenses, which would continuously deliver proteins if worn by the patient (Figures 17 & 18).



In summary, we have designed and constructed plasmids for the expression of GSR-his and CH25H-his, set up a cataract model using fish lens proteins, tested the encapsulation of proteins in chitosan nanoparticles, and used computational modeling to simulate GSR release from nanoparticles in eye drops. In the future, we envision purifying the his-tagged GSR and CH25H using immobilized metal ion affinity chromatography. The his-tag should allow our desired protein to bind to metal ions, separating from other cellular content. Furthermore, since we only tested the storage and release conditions of BSA-containing nanoparticles, we wish to repeat these tests using nanoparticles containing GSR and CH25H in the future. It may also be helpful to test the activity of the enzymes after UV exposure. Since the proteins may be exposed to UV light during the synthesis of contact lens, they may lose their enzymatic activity. Further work is necessary, but non-invasive delivery of protein drugs to the lens is a promising approach to prevent and treat cataracts.
Response to Reviewers
A transcript of the reviewer comments and author responses from the Live Peer Review Jamboree can be found here: Taipei American School Response to Reviewers
References
- World Health Organization. Prevention of blindness and visual impairment. Priority eye diseases. 2015.
- Cataracts | National Eye Institute [Internet]. Nei.nih.gov. 2017 [cited 28 February 2017]. Available from: https://nei.nih.gov/eyedata/cataract
- Segre L. How Much Does Cataract Surgery Cost? [Internet]. All About Vision. 2017 [cited 28 February 2017]. Available from: http://www.allaboutvision.com/conditions/cataract-surgery-cost.htm
- Truscott RJ. Age-related nuclear cataract—oxidation is the key. Experimental eye research. 2005 May 31;80(5):709-25.
- Giorgio M, Trinei M, Migliaccio E, Pelicci PG. Hydrogen peroxide: a metabolic by-product or a common mediator of ageing signals?. Nature reviews Molecular cell biology. 2007 Sep 1;8(9):722-8.
- Giblin FJ. Glutathione: a vital lens antioxidant. Journal of Ocular Pharmacology and Therapeutics. 2000 Apr;16(2):121-35.
- Makley LN, McMenimen KA, DeVree BT, Goldman JW, McGlasson BN, Rajagopal P, Dunyak BM, McQuade TJ, Thompson AD, Sunahara R, Klevit RE. Pharmacological chaperone for α-crystallin partially restores transparency in cataract models. Science. 2015 Nov 6;350(6261):674-7.
- Gan Q, Wang T. Chitosan nanoparticle as protein delivery carrier—systematic examination of fabrication conditions for efficient loading and release. Colloids and Surfaces B: Biointerfaces. 2007 Sep 1;59(1):24-34.
- Cvekl A, Ashery-Padan R. The cellular and molecular mechanisms of vertebrate lens development. Development. 2014 Dec 1;141(23):4432-47.
- Mello CM, Arcidiacono S, Garvey M, Gerrard J, Healy J, Soares J, Vasudevamurthy M, Wong K. Identification of Important Process Variables for Fiber Spinning of Protein Nanotubes Generated from Waste Materials. ARMY NATICK SOLDIER RESEARCH DEVELOPMENT AND ENGINEERING CENTER MA; 2012 Jan 11.
- OcluVet [Internet]. Ocluvet.com. 2017 [cited 28 February 2017]. Available from: http://www.ocluvet.com
- Wilson T, Aeschlimann R, Tosatti S, Lorenz KE. Defining ‘Fresh’Corneal Tissue for Utilization in Determining Human Cornea Coefficient of Friction Values. Investigative Ophthalmology & Visual Science. 2014 Apr 30;55(13):1506-.
- Fraunfelder FT, Fraunfelder Jr FW, Chambers WA. Clinical Ocular Toxicology: Drug-Induced Ocular Side Effects. Elsevier Health Sciences; 2008 Jun 12.
- Behl G, Iqbal J, O’Reilly NJ, McLoughlin P, Fitzhenry L. Synthesis and characterization of poly (2-hydroxyethylmethacrylate) contact lenses containing chitosan nanoparticles as an ocular delivery system for dexamethasone sodium phosphate. Pharmaceutical research. 2016 Jul 1;33(7):1638-48.
- Posner M. A comparative view of alpha crystallins: the contribution of comparative studies to understanding function. Integrative and comparative biology. 2003 Aug 1;43(4):481-91.
- Gaudana R, Ananthula HK, Parenky A, Mitra AK. Ocular drug delivery. The AAPS journal. 2010 Sep 1;12(3):348-60.
- Patel A, Cholkar K, Agrahari V, Mitra AK. Ocular drug delivery systems: an overview. World journal of pharmacology. 2013;2(2):47.
- Cholkar K, Patel SP, Vadlapudi AD, Mitra AK. Novel strategies for anterior segment ocular drug delivery. Journal of Ocular Pharmacology and Therapeutics. 2013 Mar 1;29(2):106-23.
- De Salamanca AE, Diebold Y, Calonge M, García-Vazquez C, Callejo S, Vila A, Alonso MJ. Chitosan nanoparticles as a potential drug delivery system for the ocular surface: toxicity, uptake mechanism and in vivo tolerance. Investigative ophthalmology & visual science. 2006 Apr 1;47(4):1416-25.
- de Campos AM, Diebold Y, Carbalho EL, Sánchez A, José Alonso M. Chitosan Nanoparticles as New Ocular Drug Delivery Systems: In Vitro Stability, in Vivo Fate, and Cellular Toxicity. Pharmaceutical Research. 2005 Jun 1;22(6):1007-.
Supporting Information
Supporting information for this work can be found here: Supplementary Information