iGEM REPORT: Overcoming Adversities Using DIYBIO and Open Science: Open Lab Equipment and Expression Vector System Validation in Chlamydomonas reinhardtii
Note: This iGEM Report was submitted to the PLOS iGEM Realtime Peer Review Jamboree, and has not undergone formal peer review by any of the PLOS journals. We welcome your comments on this work.
Overcoming Adversities Using DIYBIO and Open Science: Open Lab Equipment and Expression Vector System Validation in Chlamydomonas reinhardtii
Fabio N. Mello (2) *, João Vitor Dutra Molino (1), Tiago Lubiana Alves (2), Livia S. Ferreira-Camargo (1), Miguel A. Croce, Allan Tanaka (1), Felipe X. Buson (2), Pedro de Freitas Ribeiro (2), Antony B. Campos-Salazar (1), Eduardo Padilha Antonio (3), André Schraider Maizel (2), Viviane M. Siratuti (2), Bruno R. Arico (2), Mireia Recio-Mitter (2), Cauã Westmann (4), Victor N. de Jesus (5), Edmar R. de Oliveira (2), Claudia Costa (2), Samarina R. Wlodarczyk (1), Raquel de Souza Lima (2), João C. M. Carvalho (1)
- University of São Paulo, Faculty of Pharmaceutical Sciences, Department of Biochemical and Pharmaceutical Technology, São Paulo, Brazil
- University of São Paulo, São Paulo, Brazil
- Federal University of São Paulo, São Paulo, Brazil
- University of São Paulo, Faculty of Medicine, Department of Cell and Molecular Biology, Ribeirão Preto, Brazil
- State University of São Paulo “Júlio de Mesquita Filho”, Assis, Brazil
*Corresponding author: Fabio N. Mello ([email protected])
Author Contributions
Conceptualization: JVDM VMS CC RSL
Investigation: JVDM TLA LSFC AT FXB PFR ABCS EPA VMS FNM BRA MRM CW VNJ ERO SRW
Methodology: JVDM TLA ABCS ASM VMS BRA MRM
Formal Analysis: ASM BRA JVDM ABCS
Writing – original draft: FNM
Writing – review & editing: FNM JVDM ABCS MRM VNJ EPA ERO
Funding acquisition: JVDM TLA LSFC MAC AT FXB PFR ABCS EPA VMS FNM BRA MRM ERO CC SRW RSL
Supervision: JVDM LSFC JCMC
Abstract
In this project, we propose the use of Do-it-Yourself Biology (DIYBio) and Open Science concepts and tools to characterize a synthetic plasmid construct for protein expression and secretion in Chlamydomonas reinhardtii, exploring the possibilities of both approaches in providing solutions to the problems faced by scientists in emerging countries. The project as a whole was inserted in the context of the 2016 International Genetically Engineered Machine (iGEM) competition and is aligned with its effort to promote science democratization.
Financial Disclosure
The project here described was funded by Merck KGaA, Thermo Fisher Scientific, the University of São Paulo, Hardware Livre USP, Integrated DNA Technologies, Lotan, MathWorks, SnapGene, New England Biolabs and through our online public crowdfunding campaign. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing Interests
The authors declare that no competing interests exist.
Data Availability
All data related to the work described in this document is fully available without restrictions at http://2016.igem.org/Team:USP_UNIFESP-Brazil
Introduction
It is no well-kept secret that the current way academic science is done across the globe is, to say the least, in deep trouble. Classical peer-review processes seem to be less and less effective, paywalls keep crucial knowledge away from those who need it, and funding problems hamper scientific progress and push towards articles that merely aim to be published, not necessarily contributing to the field. However, science is far from being helplessly doomed. With scientific progress coming faster and faster, new alternatives and solutions to academia have been sprouting everywhere. Open Science, DIYBio, Synthetic Biology, Biohacking and many other independent movements have been taking a different approach at these problems, with the effectiveness of the solutions yet to be determined for such recent changes.[2][32][33][35][37]
The International Genetically Engineered Machine competition (iGEM) is an annual synthetic biology competition that has been bringing together undergraduate and graduate students from all over the world since its third edition in 2005.[2][32][34][36] It notably stands up for open, accessible and transparent science, which quickly drew our team, USP-UNIFESP Brazil, towards this year’s competition. Hailing from an emerging country and a region ravaged by social inequality, these ideals resonate well with the problems faced in such places by most scientists and provide an opportunity for those discontent with this establishment to try and change it. iGEM is a competition marked by teams striving in the face of adversity and circumventing problems in an unforeseen way in order to accomplish their goals,[32][34][36] so it’s only natural that teams from developing regions are a growing presence in every year’s edition.
Our project was conceived as a novel application of the modular characteristics of spider silk, in an attempt to make a biomatrix with immobilized proteins[13]-[17]. We decided to use a different model organism as an expression system, the microalgae Chlamydomonas reinhardtii , and so had to manage to not only design the expression of complex proteins as spider silk, a challenge met by other research groups in the area[5][31], but also validate a novel modified-plasmid vector responsible for the expression and secretion of said proteins. The bumpy road of taking part in iGEM 2016 was undoubtedly a huge scientific experience for all of our team members, as we are sure it was for all other teams. Most importantly, we think our greatest gain might have been precisely strengthening those ideals of open, accessible and transparent science.
Materials and Methods
Expression vector and fluorescent protein
The expression vector used for experiments on C. reinhardtii used pBluescript II plasmid as backbone and, based on recent findings, underwent a series of modifications to add functional cassettes (Figure 1, discussed below). Briefly, the promoter region was formed by two elements: (i) the heat shock protein 70 promoter (hsp70), which demonstrated to be induced under light induction.[11] (ii) three introns of the rbsc2 gene spanned on the vector which proved enhanced transgenic expression.[3] The downstream cassettes fused the bleomycin resistance gene acting as selection marker, a signal peptide from aryl sulphatase 1 gene (ARS1) to facilitate secretion and a self-cleavage peptide from Foot and Mouth Disease Virus to split the gene of interest from the rest of proteins. This combination showed suitable activity to recover proteins in supernatant fractions.[42] The gene of interest was mCherry gene, a fluorescent protein previously described.[9] The publication of the expression plasmid is still pending by the corresponding research group.
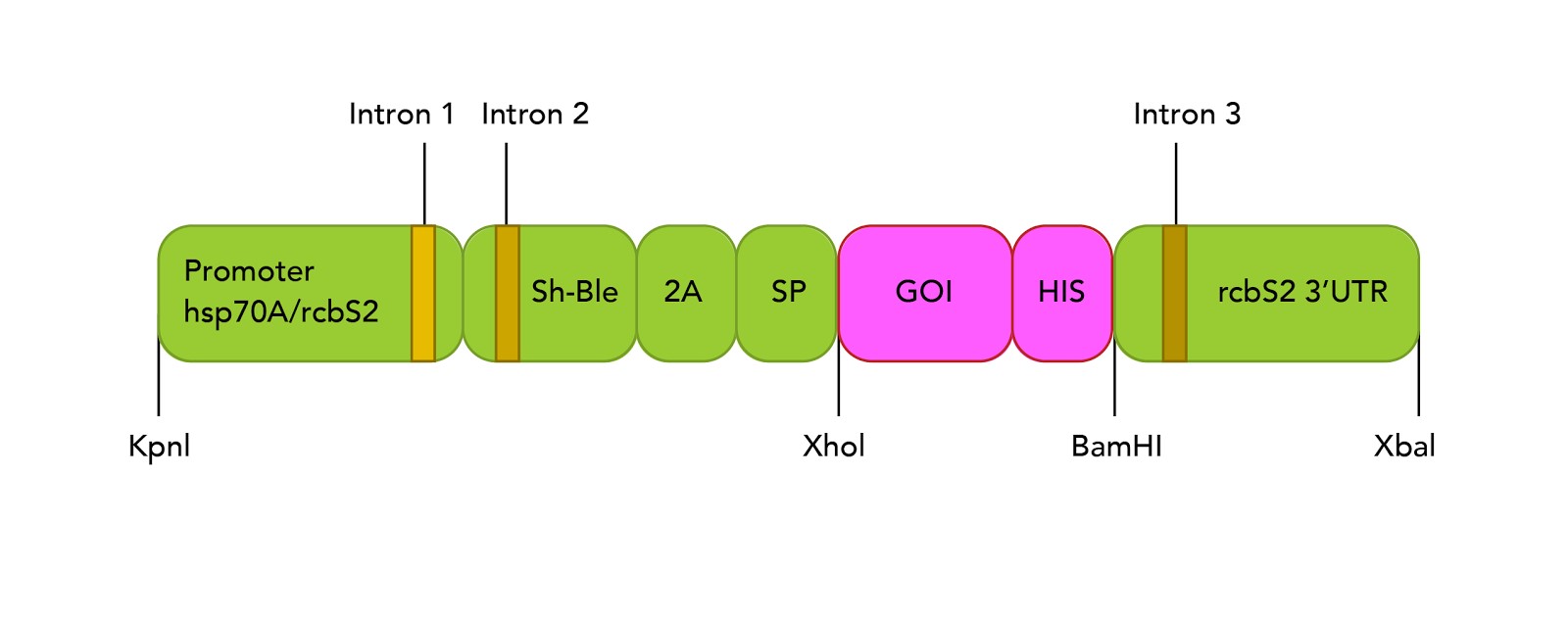
Plasmid preparation for C. reinhardtii transformation
Plasmid isolation was based on the alkaline lysis method[39] and DNA purifications were carried out following manufacturer’s recommendation for QIAquick Gel Extraction Kit (Qiagen Sciences Inc., Germantown, Maryland, USA). Polymerase chain reactions (PCR) were performed with 2X Q5 DNA polymerase (New England Biolabs, MA, USA) or home-prepared X7 Pfu DNA polymerase,[41] sufficient DNA amount and DEPC water to 25µl.
Gel electrophoresis were run on 1% agarose gels prepared with 1X sodium borate buffer at 5-35V per gel centimeter during 1-1h30, as described elsewhere.[40] Samples were composed of 4 µL of DNA template, 1 µL 10X Loading Buffer (New England Biolabs, MA, USA) and 1 µL 5X Gel Red dye (New England Biolabs, Ipswich, MA). DNA digestions were performed with 10U/µl of appropriate endonuclease(s), 1X CutSmart Buffer (New England Biolabs, Ipswich, MA), 3.5-8 µg of template DNA and DEPC water to 50 µL. Digestions were performed at 37ºC for 3 hours. Ligation reaction were performed with T4 DNA ligase, 10X T4 DNA Ligase Buffer (New England Biolabs, Ipswich, MA), DNA vector and insert in a 1:3 or 1:5 ratio and DEPC water to 20 µL. Reactions prepared at room temperature were used immediately for transformation, or left incubated at 16ºC overnight and inactivated at 65ºC for 10 min.
The expression vector containing mCherry coding sequence was used for further transformations on algae. Electroporation of C. reinhardtii were performed with 250 µL of competent cells (3-6 x 10^8 cells/ml) and, at least, 500 ng of template plasmid on 0.4 cm gap cuvettes (BIO-RAD, Hercules, CA) at 8.0 KV/cm and capacitance of 25 µF. Cells were incubated for 18 hours in 10 ml of TAP supplemented with 40mM sucrose, and then were plated on two plates of TAP agar, each supplemented with 5 or 10 µg/mL of zeocin.
Further details on protocols and lab notes can be found at http://2016.igem.org/Team:USP_UNIFESP-Brazil/Notebook
Screening of best mCherry producing colonies
Colonies were grown on a 96-well plate with 200 μL TAP media per well. Experiments were run twice at 800 RPM, 25ºC and under constant illumination of 60 micromol of photons/m^2s using different plate shakers, Agitador de MicroPlacas Analógico AM 2.4 AN (INBRAS, Jardinópolis, SP, Brazil) (mCherry, absorbance at 750 nm and chlorophyll measured once every 12 hours) and VWR INCUBATOR SHAKER-508 (Radnor, Pennsylvania, US) (mCherry measured 3 times every 12 hours, absorbance at 750 nm measured 4 times every 12 hours, chlorophyll measured once every 12 hours). Measurements were done with the following setup: mCherry fluorescence [Mode: Fluorescence Top Reading; Excitation Wavelength: 575 nm; Emission Wavelength: 608 nm; Excitation Bandwidth: 9 nm; Emission Bandwidth: 20 nm; Gain: 200 Manual; Number of Flashes: 10; Integration Time: 20 µs; Lag Time: 0 µs; Settle Time: 0 ms; Z-Position (Manual): 18141 µm], Chlamydomonas reinhardtii optical density [Mode: Absorbance; Multiple Reads per Well (Circle (filled)): 2 x 2 ;Multiple Reads per Well (Border): 750 µm; Wavelength: 750 nm; Bandwidth: 9 nm; Number of Flashes: 25; Settle Time: 0], chlorophyll fluorescence [Mode: Fluorescence Top Reading; Excitation Wavelength: 440 nm; Emission Wavelength: 680 nm; Excitation Bandwidth: 9 nm; Emission Bandwidth: 20 nm ;Gain: 100 nm; Number of Flashes: 10; Integration Time: 20 µs; Lag Time: 0 µs; Settle Time: 0 ms; Z-Position (Manual): 18141 µm].
mCherry purification
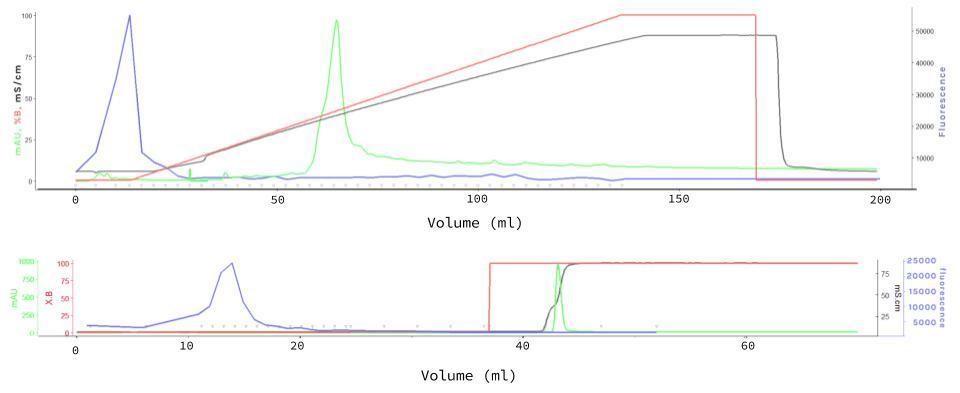
The Fast Protein Liquid Chromatography (FPLC) for mCherry purification was performed with Resource Q (6 mL) columns (GE Healthcare, Little Chalfont, UK), sodium phosphate 50 mM (pH7.5) as buffer A, sodium phosphate 50 mM (pH7.5) + 1M NaCl as buffer B, equilibration of 2 column volumes (CV), injection of 0.5 mL 40X Concentrate supernatant sample, gradient length of 20 CV, flow rate of 5 mL/min and fractionation of 5 mL to unbound and 3 mL to the rest of the method. Another more refined experiment was performed with similar conditions, in which three steps for gradient length (step 1: 3 CV, step 2: 2 CV, step 3: 5 CV), flow rate of 3 mL/min (for all steps) and fractionation of 5 mL to unbound and 1 mL on step 1, 3 mL on step 2 and 5 mL on step 3. Measurements were taken in an Infinite 200 PRO plate reader (TECAN, Männedorf, Switzerland) in the above mentioned conditions.
mCherry spectrum analysis
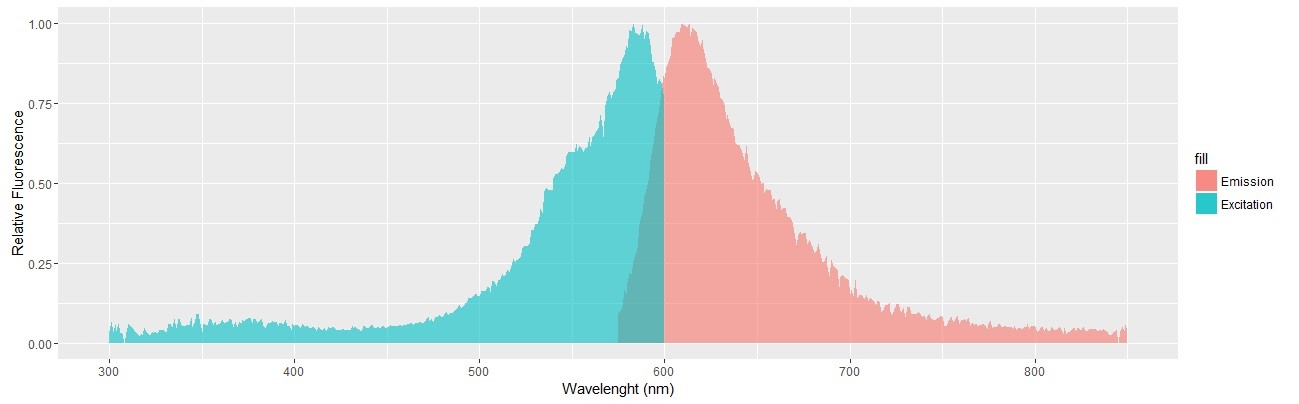
mCherry fluorescence spectrum was analyzed with the above mentioned Infinite 200 PRO plate reader with the following setup: [Mode: Fluorescence Top Reading; Excitation Wavelength Start: 300 nm; Excitation Wavelength End: 600 nm; Excitation Wavelength Step Size: 1 nm; Excitation Scan Number: 301; Emission Wavelength: 640 nm; Bandwidth (Em): 280…850: 20 nm; Bandwidth (Ex) (Range 1): 230…315: 5 nm; Bandwidth (Ex) (Range 2): 316…850: 10 nm; Gain: 200 Manual; Number of Flashes: 10; Integration Time: 20 µs; Lag Time: 0 µs; Settle Time: 0 ms; Z-Position (Manual): 18141 µm]. 100 µL of FPLC purified mCherry was used as sample.
Secretion assay
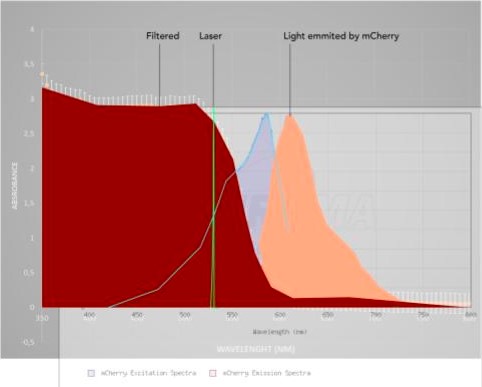
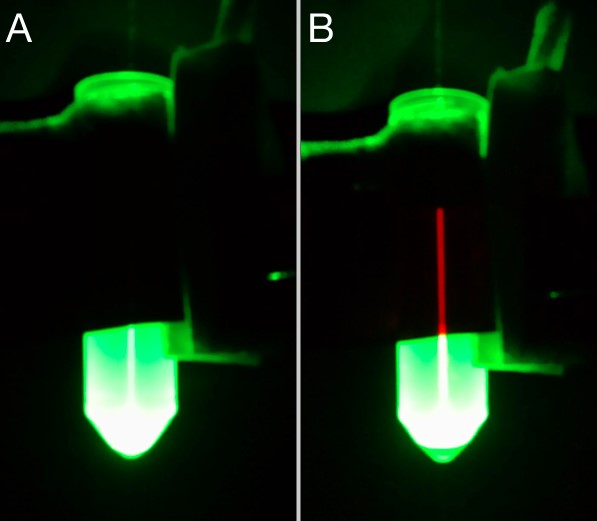
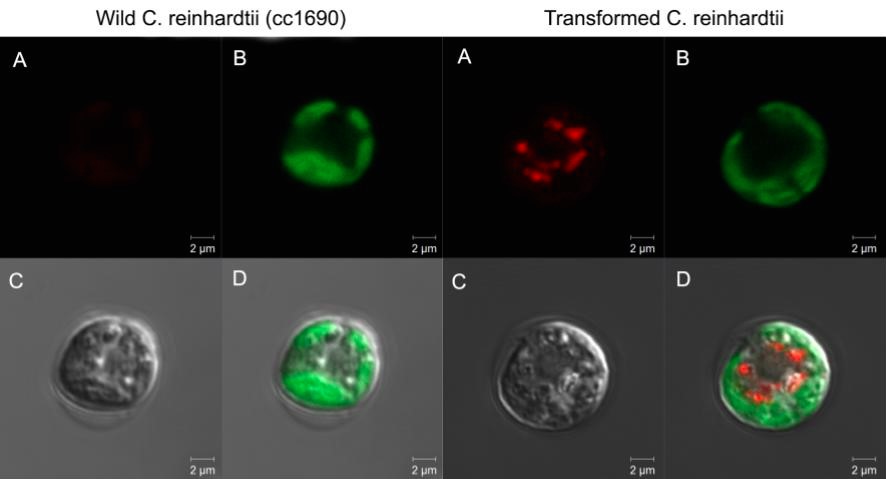
mCherry fluorescence in the supernatant was assessed using a stack of 6 Rosco E-Colour #135 light filters and a 532 nm common laser pointer. mCherry presence was also observed with a Confocal Zeiss LSM 780-NLO at 543 nm excitation and spectral detectors set for 610-650 nm range for mCherry. Chlorophyll fluorescence was detected using a 405 nm excitation wavelength and 660-700 nm wavelength.
Open access hardware and reagents
The microcentrifuge used throughout the project was built with common electronical pieces, an Arduino UNO, 3D-printed pieces and laser-cut MDF pieces. Full instructions can be found here: http://2016.igem.org/Team:USP_UNIFESP-Brazil/Hardware As other DIY materials seem to still need design improvements, we have refrained from provinding full assembly instructions as done with our centrifuge, but files for the required 3D printing, laser cutting and Arduino coding for the other equipment can be found in the same link above. Full description of DIY alternatives for reagents used, and related protocols, can be found here: http://2016.igem.org/Team:USP_UNIFESP-Brazil/Notebook.
Results and Discussion
Our team was originated from SynBio Brasil, a group of current and former students focused on spreading synthetic biology and concretizing open science, and making projects such as iGEM participations a reality. Many members of the group, some who had participated in the competition before, went on to join forces with enthusiasts from different institutes, campi and universities to comprise a team of 28 undergrad and grad students. The team size, large by iGEM standards, stemmed from the ideals held by the SynBio group of democratic and open science, and so we were joined by anyone and everyone that wanted to help, one way or another. We felt that it wasn’t right for us to turn our backs to someone who asked to join based on education level, field of study, availability for the project, or really any other reason. If the entire point of the project was to go against the way traditional science is done – elitist, held behind closed doors and with very rigid structure – repeating the same mistakes would lead us nowhere.
What would normally be seen as a liability, this multitude of team members – both in numbers as in different backgrounds – turned out to be a powerful asset when well handled. The large number meant we could segment the work and have completely different parts of the project moving forward independently, which kept the wheels rolling even if team members were unexpectedly caught busy with academic or personal matters. Normally, coordinating a team this large would be an arduous task, but we found out it is made easier when the work at hand is made simple. Technology came to our aid there: (i) Google Drive was fundamental to organize files, and also for simultaneous document editing; (ii) Slack helped for task assigning at the beginning, but it was discontinued due its low acceptance among members; (iii) online instant messaging applications allowed us to effectively communicate as a team, schedule meetings and turn a project this large into simple sizeable tasks. Furthermore, the effectiveness and near-constant availability of both our student team leader and instructor were crucial to organize the team. Emerging laboratories where synthetic biology is not the core research struggles with skilled guidance, however, a deep aid and tutoring was given by our instructors. Commitment as observed in our student team leader to conduct tasks as arranging meetings, compartmentalizing work and keeping in mind the important deadlines were essential to keep the team pace. And as horizontal as our team management might have been, and with all the positive aspects that came with it, their leadership was a great asset to the team.
The range of different areas we all came from, a quality usually not very valued, was also crucial to the project. These uniques perspectives gave us solutions that would have otherwise been ignored as we overlooked the basics and regard them as, well, too basic. Teaching other team members the core concepts of synthetic biology (and biology as a whole) simplified it even for ourselves, and in exchange the non-traditional knowledge brought in contributed in ways we couldn’t have foreseen, as in the creation our open hardware and fluorescence detection technique led by a pharmacist, an architect and a mathematician.
Synthetic production of spider silk proteins with molecular biology techniques has been attempted by other groups[5] (including previous iGEM teams[38]), but with limited success. The main difference we thought our project to have was the use of Chlamydomonas reinhardtii as our model organism. Its use for large scale production of biomaterials has been found to be very cost effective, grows rapidly and with stable lineages, with low risk of contamination by DNA-altering factors.[3][7][12] But most importantly, the naturally high GC content of its genes indicated C. reinhardtii might be better equipped to deal with the high GC content and very repetitive sequences of our target protein’s genes.[5] We then based ourselves on the expression vector (figure 1) to be used for the expression of our desired protein, and proceeded to test it with the reporter protein mCherry, expanding the usage possibilities of this red fluorescent protein from the iGEM registry. We choosed mCherry, because its photoactive spectrum is in a region of low absorption due to photosynthetic pigments inside the cells. The constructed was then successfully transformed into cc1690 lineage Chlamydomonas cells and the best producing colonies were used for further experiments (data not shown, available as previously stated). The fluorescent protein production was confirmed with fluorescence confocal microscopy, and a Fast Protein Liquid Chromatography (FPLC) (figure 2) was performed in order to purify a sample of mCherry, which then had its excitation and emission spectrums measured as shown (figure 3).
A major part of our team’s project this year revolved around overcoming basic adversities, mostly related to equipment, reactants and research budget. To put it bluntly, it basically revolved around not having a lab. Our team was very generously taken in by Professor João C. M. de Carvalho (Faculty of Pharmaceutical Sciences, University of São Paulo) in his lab, where some of us had undergraduate and graduate internships. The problem was, Professor Carvalho’s lab is focused on microalgae cultivation and optimization of biomass production processes, and lacked equipment and structure for molecular biology experiments. In fact, pretty much all of the resources we did have at our lab were either previously introduced by team members in their own academia-related projects, or equipment we were given access to in another lab, based on the immense generosity of others. We had limited access to microcentrifuges, thermocyclers, transilluminators, electrophoresis cells, electroporation systems, and many other lab materials, fundamental for molecular biology and synthetic biology experiments. The project – which was initially focused solely on the expression of spider silk-immobilized endolysins – quickly became about making solutions for our problems and holding up the idea that, despite the lack of resources, we could still do science. By the end, our project and Jamboree presentation were very heavily focused on our open hardware projects and DIY alternatives for our needed equipment (figure 4), reactants and protocols . Those jerry-rigged solutions, initially meant to be just a “quick-fix” to get basic experiments going, quickly became more and more important each step of the way. So much so, that team members themselves started dedicating more and more time to finding, testing and refining these alternatives, as well as spreading this new-found knowledge outside of our own lab, in hopes to help someone else in the same situation.

In the intent of spreading such knowledge, we have compiled all the information on our DIY equipment and made it available in our website, as referenced in the Materials and Methods section of this report. As a resource that rose up as common alternatives failed us, we are compelled to recommend it and provide free access for anyone and everyone who might find it of use. After all, one of its major benefits for us was it’s vastly reduced cost, comprised entirely of the cost of basics pieces and materials. The equipment worked smoothly for us as any other commercial alternative would have, and empowered us with an unmatched freedom to run our experiment. We were bound to have initial design flaws, as any attempt of new hardware creation, but we are proud to have fixed the noticed problems; we have designed and implemented two updates for our centrifuge in the span of six months, a rate unmatched by commercial brands. Furthermore, this open design was not only of benefit solely for us, but also for the entire community. We not only profusely recommend the DIY hardware we used, but also urge other researchers to embrace it and improve it as needed. We are unaware of any current design flaws, but conscious that there likely is vast room for improvements, and we feel we are luckily graced with the opportunity to see it improved and shared by others, spreading the creative and collaborative ideals that initially drove the conception of this project and continue to steer us.
A very relevant example of such new-found solutions was the DIY method developed to detect the secretion of mCherry by our C. reinhardtii cassette. Based off of the excitation/emission spectra measured, we used a stack of 6 photografic red filters and a common green laser pointer to screen for fluorescence in the supernatant, after centrifugation of the samples. The stacked filtered film absorption spectrum was measured (figure 5) to make sure it blocked the wavelength of the laser, but not the measured wavelength of the mCherry emission. So, by shining the laser through the sample and placing the filter in front, we would be able to isolate the desired fluorescence, as shown (figure 6). This allowed us to confirm that the signal peptide of our construct worked as expected, while being faster and cheaper than traditional methods and without sacrificing reliability. In interest of a double confirmation, confocal fluorescence microscopy was also used to detect mCherry fluorescence (figure 7). Although not pursuit during the project, mCherry fluorescence could have being quantified employing a similar approach we used for the interlab experiments. Basically, by setting a similar experiment layout, taking pictures in fixed set ups, and comparing brightness using ImageJ (See Interlab; Macroscopy analysis). Other fluorescence proteins might be used in C. reinhardtii or other organisms, needing only a suitable excitation source and filter. GFP detection is readily observable using a UVB lamp and a green filter. Alternatively, 390 nm blue LEDs could be used, provided they deliver enough light power and an efficient filter is in place. Table 1 presents a set of possible LEDs as source light and suitable filters film may be found in companies as Rosco with their Roscolux line.

Conclusions
The synthetic production of spider silk proteins as a biomaterial for immobilized enzymes remains, unfortunately, unachieved by the end of our 2016 project. As shown, Chlamydomonas reinhardtii model holds potential for a future project along this line, now helped by the construction and characterization of our DNA cassette for protein expression and secretion.
Following iGEM’s lead, all of our completed DIY-projects – both original designs and projects adapted from other sources and rightfully credited – have been fully uploaded in our iGEM wiki with accessible instructions for anyone who may need it in the future. Furthermore, we took part in the Brazilian FabLearn Conference in São Paulo, as well as, presented our hardware projects at FAZ, a maker festival organized by Red Bull, and even held our own public hackathon at the University of São Paulo, with the aim to spread the word about open hardware and collaborative science. After all, we would do nothing to solve the problems we encountered if we kept the solution locked away. With the rise of the community labs such as GenSpace, and movements like biohacking or DIYBio, this kind of open access low-cost equipment may prove itself to be an affordable and reliable option, with an added benefit: due to its decentralized and collaborative nature, these equipment can be a massive instrument to tear down the divisions between academia and non-academia, bringing scientific knowledge and tools to the public.[2][32][33][35][37]
After our iGEM Jamboree experience, and meeting the other two Brazilian teams which participated this year, an idea to make our own regional Jamboree in 2017 sprouted among us. Brazil, as many other countries in Latin America, is still stuck with the same old scientific scenario, which is not only exclusionary but also limiting of itself. Scientific progress is done in tiny steps in our country, and we believe that supporting projects such as iGEM participations is crucial to change that. We realized that many teams in Brazil are not formed due to the present adversities, despite having interested students and researchers, and so we would all like to organize an event to help foster new ideas for new teams, and help each other to keep going. As of now, our own regional Jamboree is still being planned, but by bringing Brazilian iGEM team members and enthusiasts together, it is bound to lead to great things in the future.
Acknowledgments
We would like to thank the Professors, friends, companies and institutes that helped making this project possible. We are thankful to Lukasz Kowalik (Merck KGaA), Marlene Lunardi (Thermo Fisher Scientific), Otto Heringer and Danilo Zampronio (Lotan), Research Ideas and Outcomes (Journal RIO), São Paulo Research Foundation (FAPESP), Núcleo de Empreendedorismo da USP (NEU), Professors Ricardo Pinheiro de Souza Oliveira, Ph.D., Gisele Monteiro, Ph.D., Mario Hiroyuki Hirata, Ph.D. and Professor Adalberto Pessoa Junior, Ph.D. (University of São Paulo/Pharmacy School), Professors Luiziana Ferreira da Silva, Ph.D., Professor José Gregório Cabrera Gomez, Ph.D., Professor Márcio Bertacine Dias, Ph.D. and Mario Costa Cruz, M.Sc. (University of São Paulo/Biomedical Sciences Institute), Professors Alexandre Bruni-Cardoso, Ph.D. and William Cenens, Ph.D. (University of São Paulo/Institute of Chemistry), Professor Eduardo de Senzi Zancul, Ph.D. (University of São Paulo/Polytechnic School), Professor Stephen Patrick Mayfield, Ph.D. (University of California, San Diego/Biological Sciences Division), Professors Edislane B. de Souza, Ph.D., Dario A. Palmieri, Ph.D., Pedro O. Neto, Ph.D. (São Paulo State University, Assis/Dep. of Biotechnology), Professor Ana Maria Pires, Ph.D. and Airton Germano Bispo Júnior, M.Sc. (São Paulo State University, Presidente Prudente/Dep. of Chemistry and Biochemistry), Ph.D. and Elibio Leopoldo Rech Filho, Ph.D. (Brazilian Agricultural Research Corporation (EMBRAPA – Genetic Resources & Biotechnology)) and Professor Carlos Gustavo Nunes da Silva, Ph.D. (Federal University of Amazonia UFAM).
Response to Reviewers
A transcript of the reviewer comments and author responses from the Live Peer Review Jamboree can be found here: USP-UNESP-UNIFESP Response to Reviewers
References
- Molino JVD, Alves TL, Ferreira-Camargo LS, Croce MA, Tanaka A, Buson FX, Ribeiro PF, Campos-Salazar AB, Antonio EP, Maizel AS, Siratuti VM, Costa C, Wlodarczyk SR, Lima RS, Mello FN, Mayfield SP, Carvalho JCM (2016) Research Ideas and Outcomes 2: e9342 DOI: https://doi.org/10.3897/rio.2….
- Landrain T, Meyer M, Perez AM, Sussan R (2013) “Do-it-yourself biology challenges and promises for an open science and technology movement” Systems and Synthetic Biology, 7 (3): 115–126. DOI: 10.1007/s11693-013-9116-4
- Eichler-Stahlberg A, Weisheit W, Ruecker O, Heitzer M (2009) “Strategies to Facilitate Transgene Expression in Chlamydomonas reinhardtii.” Planta 229 (4): 873–883. DOI: 10.1007/s00425-008-0879-x
- Fuhrmann M, Oertel W, Hegemann P (1999) “A Synthetic Gene Coding for the Green Fluorescent Protein (GFP) Is a Versatile Reporter in Chlamydomonas reinhardtii” The Plant Journal: for cell and molecular biology 19(3): 353–361. DOI: 10.1046/j.1365-313X.1999.00526.x
- Lewis, RV (2006) “Spider Silk: Ancient Ideas for New Biomaterials” Chemical Reviews 106 (9): 3762–3774. DOI: 10.1021/cr010194g
- Lumbreras V, Stevens DR, Purton S (1998) “Efficient Foreign Gene Expression in Chlamydomonas reinhardtii Mediated by an Endogenous Intron” The Plant Journal: for cell and molecular biology 14 (4): 441–447. DOI: 10.1046/j.1365-313X.1998.00145.x
- Mayfield SP, Franklin SE, Lerner RA (2003) “Expression and Assembly of a Fully Active Antibody in Algae” Proceedings of the National Academy of Sciences 100 (2): 438–442. DOI: 10.1073/pnas.0237108100
- Mayfield SP, Manuell AL, Chen S, Wu J, Tran M, Siefker D, Muto M, Marin-Navarro J (2007) “Chlamydomonas reinhardtii Chloroplasts as Protein Factories” Current opinion in biotechnology 18 (2): 126–133. Print.
- Rasala BA, Lee PA, Shen Z, Briggs SP, Mendez M, Mayfield SP (2012) “Robust Expression and Secretion of Xylanase1 in Chlamydomonas reinhardtii by Fusion to a Selection Gene and Processing with the FMDV 2A Peptide” PloS one 7 (8): e43349. DOI: http://dx.doi.org/10.1371/jour…
- Rosenberg JN, Oyler GA, Wilkinson L, Betenbaugh MJ (2008) “A Green Light for Engineered Algae: Redirecting Metabolism to Fuel a Biotechnology Revolution” Current Opinion in Biotechnology 19 (5): 430–436. Print.
- Schroda M, Blöcker D, Beck CF (2000) “The HSP70A Promoter as a Tool for the Improved Expression of Transgenes in Chlamydomonas .” The Plant Journal: for cell and molecular biology 21 (2):: 121–131. DOI: 10.1046/j.1365-313x.2000.00652.x
- Wijffels RH, Kruse O, Hellingwerf KJ (2013) “Potential of Industrial Biotechnology with Cyanobacteria and Eukaryotic Microalgae” Current Opinion in Biotechnology 24 (3): 405–413. DOI: http://dx.doi.org/10.1016/j.co…
- Andersson M, Chen G, Otikovs M, Landreh M, Nordling K, Kronqvist N, Westermark P, Jörnvall H, Knight S, Ridderstråle Y, Holm L, Meng Q, Jaudzems K, Chesler M, Johansson J, Rising A (2014) “Carbonic Anhydrase Generates CO2 and H+ That Drive Spider Silk Formation Via Opposite Effects on the Terminal Domains” PLoS Biology 12 (8): e1001921. DOI: 10.1371/journal.pbio.1001921
- Ayoub N, Garb J, Tinghitella R, Collin M, Hayashi C (2007) “Blueprint for a High-Performance Biomaterial: Full-Length Spider Dragline Silk Genes” PLoS ONE 2 (6): e514. DOI: 10.1371/journal.pone.0000514
- Blüm C, Nichtl A, Scheibel T (2013) “Spider Silk Capsules as Protective Reaction Containers for Enzymes” Advanced Functional Materials 24 (6): 763‑768. DOI: 10.1002/adfm.201302100
- Garb JE, Ayoub NA, Hayashi CY (2010) “Untangling spider silk evolution with spidroin terminal domains” BMC Evolutionary Biology 10 (1): 243. DOI: 10.1186/1471-2148-10-243
- Hardy J, Römer L, Scheibel T (2008) “Polymeric materials based on silk proteins” Polymer 49 (20): 4309‑4327. DOI: 10.1016/j.polymer.2008.08.006
- Heitzer M, Zschoernig B (2007) “Construction of modular tandem expression vectors for the green alga Chlamydomonas reinhardtii using the Cre/lox-system” BioTechniques 43 (3): 324‑332. DOI: 10.2144/000112556
- Hojckova K, Stano M, Klucar L (2013) “phiBIOTICS: catalogue of therapeutic enzybiotics, relevant research studies and practical applications” BMC Microbiology 13 (1): 53. DOI: 10.1186/1471-2180-13-53
- Khan AA, Alzohairy M (2010) “Recent Advances and Applications of Immobilized Enzyme Technologies: A Review” Research Journal of Biological Sciences 5 (8): 565‑575. DOI: 10.3923/rjbsci.2010.565.575
- Kluge J, Rabotyagova O, Leisk G, Kaplan D (2008) “Spider silks and their applications” Trends in Biotechnology 26 (5): 244‑251. DOI: 10.1016/j.tibtech.2008.02.006
- Lauersen K, Berger H, Mussgnug J, Kruse O (2013) “Efficient recombinant protein production and secretion from nuclear transgenes in Chlamydomonas reinhardtii” Journal of Biotechnology 167 (2): 101‑110. DOI: 10.1016/j.jbiotec.2012.10.010
- León-Bañares R, González-Ballester D, Galván A, Fernández E (2004) “Transgenic microalgae as green cell-factories.” Trends in biotechnology 22 (1): 45‑52. DOI: 10.1016/j.tibtech.2003.11.003
- Liese A, Hilterhaus L (2013) “Evaluation of immobilized enzymes for industrial applications” Chemical Society Reviews 42 (15): 6236. DOI: 10.1039/c3cs35511j
- Merabishvili M, Pirnay J, Verbeken G, Chanishvili N, Tediashvili M, Lashkhi N, Glonti T, Krylov V, Mast J, Parys LV, Lavigne R, Volckaert G, Mattheus W, Verween G, Corte PD, Rose T, Jennes S, Zizi M, Vos DD, Vaneechoutte M (2009) “Quality-Controlled Small-Scale Production of a Well-Defined Bacteriophage Cocktail for Use in Human Clinical Trials” PLoS ONE4 (3): e4944. DOI: 10.1371/journal.pone.0004944
- Monier M (2013) “Immobilization of β-galactosidase from Escherichia coli onto modified natural silk fibers” Journal of Applied Polymer Science 130 (4): 2923‑2931. DOI: 10.1002/app.39475
- O’Flaherty S, Coffey A, Meaney W, Fitzgerald GF, Ross RP (2005) “The Recombinant Phage Lysin LysK Has a Broad Spectrum of Lytic Activity against Clinically Relevant Staphylococci, Including Methicillin-Resistant Staphylococcus aureus” Journal of Bacteriology 187 (20): 7161‑7164. DOI: 10.1128/jb.187.20.7161-7164.2005
- Rasala B, Mayfield S (2011) “The microalga Chlamydomonas reinhardtii as a platform for the production of human protein therapeutics” Bioengineered Bugs 2 (1): 50‑54. DOI: 10.4161/bbug.2.1.13423
- Sheldon RA, van Pelt S (2013) “Enzyme immobilisation in biocatalysis: why, what and how.” Chemical Society reviews 42 (15): 6223‑6235. DOI: 10.1039/c3cs60075k
- Tokareva O, Jacobsen M, Buehler M, Wong J, Kaplan D (2014) “Structure–function–property–design interplay in biopolymers: Spider silk” Acta Biomaterialia 10 (4): 1612‑1626. DOI: 10.1016/j.actbio.2013.08.020
- Yang Y, Qian Z, Zhong J, Xia X (2016) “Hyper-production of large proteins of spider dragline silk MaSp2 by Escherichia coli via synthetic biology approach” Process Biochemistry 51 (4): 484‑490. DOI: 10.1016/j.procbio.2016.01.006
- Vilanova C, Porcar M (2014) “iGEM 2.0 refoundations for engineering biology” Nature Biotechnology 32: 420–424. DOI: 10.1038/nbt.2899
- Vachelard J, Gambarra-Soares T, Augustini G, Riul P, Maracaja-Coutinho V (2016) ”Guide to Scientific Crowdfunding” PLoS Biol 14(2): e1002373. doi:10.1371/journal.pbio.1002373
- Tsui J, Meyer AS, Yeoman K (2016) “Modular projects and ‘mean questions’: best practices for advising an International Genetically Engineered Machines team” FEMS Microbiol Lett 363 (14): fnw141. DOI: https://doi.org/10.1093/femsle…
- Jefferson C, Lentzos F, Marris C (2014) “Synthetic biology and biosecurity: challenging the ‘myths’” Frontiers in Public Health 2: 115. doi: 10.3389/fpubh.2014.00115
- Balmer AS, Bulpin KJ (2013) “Left to their own devices: Post-ELSI , ethical equipment and the International Genetically Engineered Machine (iGEM) Competition” BioSocieties 8: 311. DOI:10.1057/biosoc.2013.13
- 2016 Keulartz J, van den Belt H (2016) “DIY-Bio – economic, epistemological and ethical implications and ambivalences” H. Life Sci Soc Policy (2016) 12: 7. doi:10.1186/s40504-016-0039-1
- http://2015.igem.org/Team:UCLA
- Sambrook J., Fritsch E. F., Maniatis T., (1989) “Molecular Cloning: A Laboratory Manual” Cold Spring Harbor Laboratory, Cold Spring Harbor, NY.
- Brody J. R., Kern S. E. (2004) “Sodium boric acid: a Tris-free, cooler conductive medium for DNA electrophoresis” BioTechniques 36: 214-216
- Morten HH Nørholm (2010) “A mutant Pfu DNA polymerase designed for advanced uracil-excision DNA engineering” BMC Biotechnology 10(21) DOI:10.1186/1472-6750-10-21
- Rasala B.A., Barrera D.J., Ng J., Plucinak T.M., Rosenberg J.N., Weeks D.P., et al (2013) “Expanding the spectral palette of fluorescent proteins for the green microalga Chlamydomonas reinhardtii.” Plant J 74(4):545–56