iGEM REPORT: Combining the Biological Unnatural Amino Acid (UAA) Orthogonal Genetical System with Logic “AND” Gate Gene Circuit to Kill Bladder Cells Precisely
Note for Reviewers
This work has been submitted for open review as an iGEM Report.
Reviewers, please consider the following questions when reviewing the report:
1. Does the submission accurately and clearly report the experiment carried out?
2. Does the report adhere to the PLOS Data Policy?
3. Is the report presented correctly and well written?
See iGEM Reviewer Instructions for more information.
Please leave your review as a comment below the Report.
Combining the Biological Unnatural Amino Acid (UAA) Orthogonal Genetical System with Logic “AND” Gate Gene Circuit to Kill Bladder Cells Precisely
The project is completed by all members of the 2015 SZU-iGEM team from China.
Yongyi Wong(1,2), Wanxin Ye(1,3), Hao Wong (1,2), Dawei Yi (1,2), Zhiwei Zhang(1,4) , Shaopei Xie(1,5) , Kexin Li (1,4), Huilin Xie(1,4) ,Changxing Hu(1,2),Shuang Liang(1,2), Yin Xiao (1,2), Xue Chen(1,2) ,Hui Ai(1,2), Zeyu Miao(1,2), Fang Shu(1,2), Zhipeng Qiu(1,2), Zhangli Hu(6), Zhiming Cai(10), Beixin Mo(7), Weiren Huang(9), Qing Zhou(9), Jiangxin Wong(8), Weizhao Chen(8)*
- Shenzhen University Team (SZU-China) for the International Genetically Engineered Machine Competition (iGEM)
- College of Life Science , Shenzhen University
- College of Business Administration, Shenzhen University
- Institute of Advanced Study, Shenzhen University
- College of Computer and Software science, Shenzhen University
- Advisor, The Dean of the College of Life Science, Shenzhen University
- Advisor, The current Vice President of College of Life Sciences, Shenzhen University
- Instructor, Professor of the College of Life Science, Shenzhen University
- Instructor, Doctor of The Second People’s Hospital of Shenzhen
- Advisor, The Director of The Second People’s Hospital of Shenzhen
Authors Contributions
- Conceptualization: W.Chen,Wong ,Q.Zhou,Y.Wong,D.Yi,H.Wong,W.Ye
- Methodology: Y.Wong,D.Yi,H.Wong, C.Hu,F.Shu
- Investigation & Collect data: W.Ye,Liang,H.Ai,Z.Miao, K.Li,H.Xie,Y.Xiao, X.Chen
- Writing—Original Draft: Y.Wong,D.Yi,H.Wong,W.Ye,F.Shu,K.Li,H.Xie
- Review & Editing: W.Chen,Q.Zhou,Y.Wong,D.Yi,H.Wong,W.Ye
- Sponsor Funding Acquisition: W.Chen,Wong,W.Ye
- Project Administration: W.Chen,Q.Zhou,Y.Wong, W.Ye
- Supervision: W.Chen, Q.Zhou
Abstract
Bladder cancer is listed as the most commonly encountered urinary carcinoma in China. This study tries to construct a gene circuit based on AND GATE and Unnatural Amino Acid Orthogonal System to selectively express therapeutic gene in bladder cancer cells and kill them. hTERT and hUPII, which are cancer specific and bladder specific promoter respectively, were used to control the expression of the output gene distinctively in bladder cancer cells. The gene circuit of this study is composed of three plasmids: hUPII-AckRS, hTERT-tRNACUA and SV40-(Effector). The effector in the third plasmid can be any gene that can produce a certain kind of protein, such as Rluc, GFP, Bax or P21, based on the purpose of the project. However, any effector used can be transcripted to an mRNA with a codon being mutated to TAG, the terminator, so that it cannot be expressed in natural condition. The tRNACUA being produced by the second plasmid has an anticodon CUA, which can pair with the terminator. Ack, a kind of unnatural amino acid, should be added to the culture medium. With the help of AckRS, a kind of tRNA synthetase produced by the first plasmid, Ack can attach to the tRNA and the whole peptide chain can then be produced. Only in this way can the effector be expressed and make sure this gene circuit will not perform its function in natural condition. Using Luc as the reporter gene, the result of this study shows that the expression level of Luc in 5637, a bladder cancer cell line, is significantly higher than that in Hela, a cervical carcinoma cell line and Human Fibre Epithelial cells. Also, it proves that the circuit can only perform its function with Ack in the culture medium, indicating that this design is in high specificity and safety.
Introduction
As we all know, bladder is a vital organ in our urinary system. However, bladder cancer is listed as the most commonly encountered urinary carcinoma in China and it ranks No.2 in America. According to management goals and prognosis, the bladder cancers are classified as non-muscle-invasive, muscle-invasive, and metastatic.[1] Although methods for early diagnosis of bladder cancer have been improved, the 5-year survival rate of metastatic bladder cancer has not been changed significantly[2] According to the National Cancer Institute of the United Nation, almost 74,000 people in America will be diagnosed with bladder cancer this year and this number has been increasing for 30 years, which means more and more people will have to face bladder cancer in the future. Therefore, it’s very important for us to come out with an efficient way to cure bladder cancer.
There are many types of treatment for bladder cancer nowadays. Chemotherapy and radiation therapy, for instance, are two major types of treatments.[3] However, chemotherapy can not only kill cancer cells, but also healthy cells that grow and divide very quickly, thus creating a considerable side effects such as mouth sore and hair loss. There is also another type of treatment called targeted therapy, which can specifically target at cancer cells with no affect on healthy cells. So this year, we’re trying to construct a gene circuit based on AND GATE and Unnatural Amino Acid (UAA) Orthogonal System to selectively express therapeutic gene in bladder cancer cells to kill them specifically.
Digital logic AND gate circuits
As we all know, there many logic gates in digital circuits.Among them, AND gate is the most basic logic gate circuit. (shown in Fig.1,source:https://en.wikipedia.org/wiki/AND_gate)
It can be seen as a switch, but we need two or more keys to open it, and it’s very according with our requirements. When we need multiple factors limit, AND gate is undoubtedly the best choice, because only all of our requirements are satisfied, the switch can be opened, and our system can run normally[4].
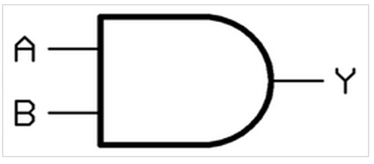 Fig.1: AND gate system schematic diagram
Fig.1: AND gate system schematic diagram
Orthogonal system
This system has no or little cross with the natural biological system which is composed of unnatural base pair, shift code, unnatural amino acid, orthogonal aaRS(aminoacyl tRNA synthetase), orthogonal RNA synthetase, promoter and the orthogonal ribosome[5].These components work corporately or separately in the organism, programming cells with new functions, and also providing new techniques for researchers.(shows in Fig.2,source: Incorporating Unnatural Amino Acids into Recombinant Proteins in Living Cells, see in reference [16])
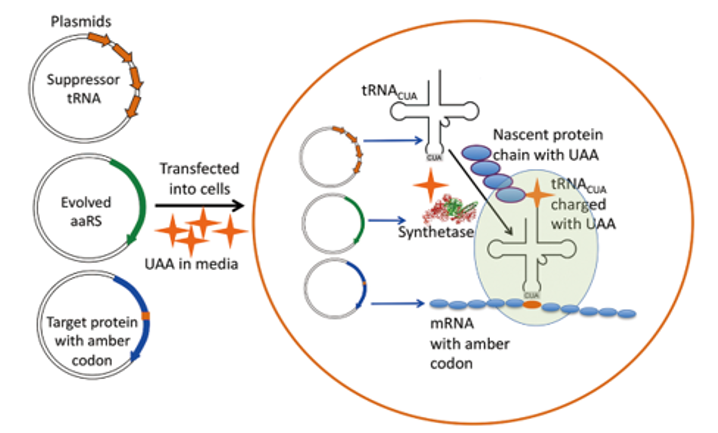
Fig. 2: Schematic of method of UAA incorporation
Plasmids encoding the suppressor tRNACUA, the evolved amino acyl-tRNA synthetase (aaRS) and the target protein with the amber codon are transfected into cells. The media covering the cells is supplemented with the UAA (orange cross). The aaRS catalyses the acylation of the suppressor tRNACUA with the UAA. When the mRNA containing the amber codon is being translated in the ribosome (light green oval), the amber codon is recognised by the tRNACUA charged with the UAA and this amino acid is added to the growing polypeptide chain.
What is UAA
UAA is short for Unnatural Amino Acids.In most organisms, there are 20 kinds of amino acids in DNA encoding, which form the vast majority of proteins in the present life.Part of the ancient bacteria (Archaea) and (eubacteria) can encoding selenium (selenocysteine) or the pyrrolysine. The amino acids which do not exist in the natural protein sequence are called UAA[6].The unnatural amino acids used in the system must be stable, and can be stable in the cell.
The UAA using in our system: Acetyllysine (ACK)
Acetyllysine(ACK) is one of unnatural amino acid and only corresponds to a special aminoacyl-tRNA(tRNA). It can be added to the protein by Semantic coding of termination codon(UAG).
Fig.3 Source:https://en.wikipedia.org/wiki/Acetyllysine,https://en.wikipedia.org/wiki/Lysine,disposed by Yongyi Wong of SZU-China 2015 iGEM team.
Fig. 3: Acetyllysine & Lysine chemical structure diagram
Use UAA as a switch to start the translation of proteins
To expand the genetic code and to lead new amino acids into the biosynthetic system, we need to identify the unnatural orthogonal codon, the tRNA with blank codon,the aaRS which can load UAA onto the tRNA specifically, the orthogonal mRNA, which can identify and highly efficient orthogonal translation, appropriate ribosome-binding site (RBS) [7]. These components need to satisfy the following conditions: (1) the only code must be recognized by the new tRNA, and has a link-coupled aaRS; (2) the aaRS/tRNA combination must be highly specific for unnatural amino acids in the host cells; (3) aaRs/tRNA pairs must be orthogonal differentiating endogenous aaRS/tRNA pairs[8].(shows in fig.4,source: Unnatural Amino Acid Mediated Protein Bioorthogonal Labeling, see reference [17] )
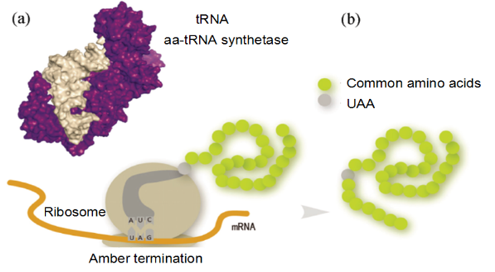
Fig. 4: Structure of tRNA/synthetase complex (a) and mechanism of genetic incorporation of unnatural amino acids (b)
Materials and Methodology
Material–Cell lines & Strain
- Tested cell lines: Bladder cancer cell line 5637 and T24, Hela cell line and HFC(Human Fiber epithelial Cell)
All of the above cell lines were kindly given by Prof. Z.Cai of The Second People’s Hospital of Shenzhen.
- Engineered construction strain: coli DH5α
The E.coli DH5α strain was kept in our iGEM laboratory in -80°C refrigerator.
Material–Plasmids
Plasmid backbone: psiCHECKTM-2, bought from Promega company.
We used psiCHECKTM-2 as the plasmid backbone to construct two plasmids and three plasmids system. The reporter of the two plasmids system was Luc, and the reporter of the three plasmids system was Rluc, in which the Luc was mutated. Exception the Luc and Rluc, we also used GFP as reporter in two plasmids and three plasmids system.

Fig.5: The two plasmids system and the three plasmids system in SZU-China 2015 iGEM team project
Methodology
We combine UAA Orthogonal genectical system with logic AND gate gene circuits to kill bladder cancer cells precisely. The following Methodology diagram was generated by Shaopei Xie of SZU-China 2015 iGEM team.

Fig.6: The methodology in SZU-China 2015 iGEM team project
The AND gate of our project
The AND gate of the team system was generated by Hao Wong of SZU-China 2015 iGEM team.(Fig.5)
The AND gate of our system has two keys, and that is two promoters, hTERT and hUPll. The former is a cancer cell specific promoter, and the latter is bladder cell specific promoter. When two keys exist at the same time and two promoters will start gene expression, the whole system can operate[9].Using AND gate to achieve specificity recognition, when it is compared with the traditional therapy, it is the biggest advantage of synthetic biology, and it’s also the basis of our project[10].
hRluc is a reporter that can produce RLUC which is a kind of luciferase. There is an amber stop codon UAG in the sequence of Rluc. When the system is verified having a function, it will produce an active hRluc.

Fig. 7: The schematic diagram of AND gate system of SZU-China project. hUPII and hTERT are specific promoters in bladder cancer cells.
The construction of Orthogonal system
The Orthogonal gene circuit system was generated by Hao Wong and Yongyi Wong of SZU-China 2015 iGEM team.(Fig.6)
In our project, when AND gate operates the tRNA as well as AckRS are desperately synthesized. The certain tRNA can identify and combine terminations codon UAG. AckRS can achieve the attachment between unnatural amino acid Ack and the tRNA. Then the tRNA charged with unnatural amino acid Ack is able to specifically promote a certain protein translation[11]. Eventually the certain mRNA can be translate successfully and output some effectors to regulate tumors growth, motility and apoptosis. The benefits of our UAA orthogonal system is that it can only working in our bladder while unable to interfere our regular genetic system.
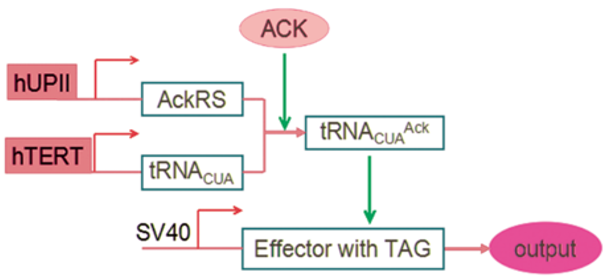
Fig. 8: The gene circuit of SZU-China Sniper system
The combination of AND gate and Orthogonal system
The combination of AND gate and Orthogonal system diagram was generated by Hao Wong of SZU-China 2015 iGEM team.(Fig.7)
2015 SZU-iGEM constructed SV40(with Enhancer) and Rluc with one codon being amber mutated in the same plasmid. This plasmid, together with two other plasmids, are inserted into the cell. Only when the three plasmids work simultaneously can our orthogonal system behave its function, specifically recognise bladder cancer cells and kill them. Our unnatural amino acid orthogonal system consists of three devices (plasmids).
(1)hUPll+AckRS(BBa_K1722007):
hUPll is a bladder-cell specific promoter. when it’s activated, AckRS, a tRNA synthetase, will be produced.
(2)hTERT+tRNA(BBa_K1722010)/ shTERT+tRNA(BBa_K1722011):
hTERT and shTERT are cancer-cell specific promoters. tRNA can be expressed out when the promoter is activated.
(3)SV40+Rluc(BBa_K1722012):
SV40 is a widely used strong promoter. Rluc is a reporter that can produce RLUC which is a kind of luciferase.
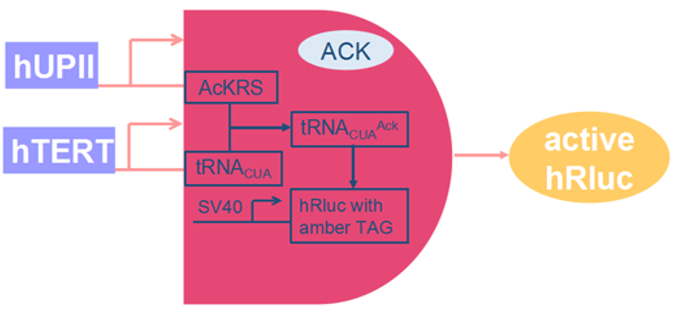
Fig. 9: The gene circuit of SZU-China Sniper system based on AND gate and Orthogonal system
Results
Renilla Luciferase assay in three plasmids system
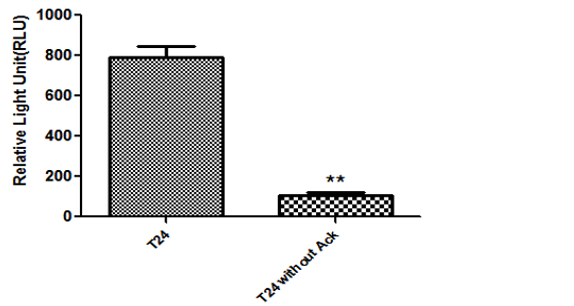
Three recombinant plasmids were generated and transiently transfected into a human bladder cancer cell line T24.
To test the working efficiency of our orthogonal system, we divided the cells into two groups. One has Ack in the culture medium and another does not.
Luciferase assays were performed using luminometer. Since Ack is essential for our system, we speculated that Renilla Luciferase (Rluc), the output gene in the circuits can be selectively expressed in Experiment group with Ack[12]. As shown in Fig. 8, the activity of Renilla Luciferase (RLUC) varied widely between the two groups, and the circuit only demonstrated significant activity in the experimental group as expected[13]. Therefore, our orthogonal system is verified to be in good condition and can work efficiently.
Fig. 10: The relative light activity of Rluc in three plasmids system.
Plasmids psiCHECKTM2-hUPII-AckRS, psiCHECKTM2-hTERT-tRNA and psiCHECKTM2-SV40-Rluc were transiently transfected into T24, a bladder cancer cell line. Ack is an essential unnatural amino acid which is short for Acetyllysine. *P-value <0.05, **P-value<0.01 and ***P-value <0.0001.
Luciferase assay in two plasmids system
hTERT and hUPII promoters are cancer specific promoter and bladder specific promoter, respectively. By combining the two promoters into the design, we supposed that the constructed circuits have the ability to selectively identify bladder cancer cells, in which telomerase and uroplakins are both expressed.
To test this hypothesis, we used the luciferase reporter gene as the output of the circuit and tested the luciferase activity in HFC, which is short for human fiber epithelial cell, Hela, a cervical carcinoma cell line and 5637, a bladder cancer cell line. Also, we reconstructed our system in two plasmids to test if two plasmids system can perform higher efficiency[13].
The result (Fig.11) shows that the activity of LUC in 5637 with Ack was about two times as high as that in 5637 without Ack and three times as high as that in Hela, while it could not be measured in HFC. We can learn from this result that this system is perfectly safe for normal cells, with no luciferase being detected in HFC. However, compared with three plasmids system, whose RLUC activity of experimental group is about 7 times as high as that of control group, the working efficiency of our orthogonal system in two plasmids is much lower[14]. So we had better construct our system in two plasmids to express higher level of therapeutic gene in bladder cancer cells and reduce its expression in other cell types to increase its specificity.
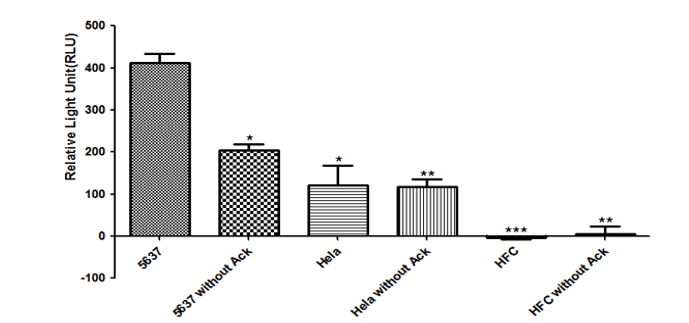
Fig. 11: The relative light activities of Luc in two plasmids system
Plasmids psiCHECKTM2-hUPII-AckRS-HSVTK-Luc and psiCHECKTM2-hTERT-tRNA-HSVTK-Luc were transiently transfected into these cell lines. 5637 is a bladder cancer cell line, Hela is a cervical carcinoma cell line and HFC is short for human fiber epithelial cell line. Ack is an essential unnatural amino acid in our system which is short for Acetyllysine. Results are shown as mean +s.e. *P-value<0.5, **P-value<0.01, and ***P-value<0.0001, relative to the 5637 group by two-tailed t-test.
Green fluorescent light measurement in two and three plasmids system
To further verifying the working efficiency of the unnatural amino acid orthogonal system, we replaced the amber mutated output gene in former plasmids to amber mutated GFP. The plasmid psiCHECKTM2-CMV-GFP was used as a positive control to monitor transfection and expression efficiency[15]. The two plasmids and three plasmids system being constructed by GFP recombinant plasmids were transiently transfected into 5637 and T24, both of which are bladder cancer cell line.
Pictures of green fluorescent light produced by target cells were taken by fluorescent microscope. From these pictures (Fig. 10), we can see there is no expression of GFP being detected in no Ack groups while the light intensities are rather high in with Ack groups no matter in T24 or 5637 cell lines. These results indicate that the output gene is expressed only in cells which have Ack in the culture medium.
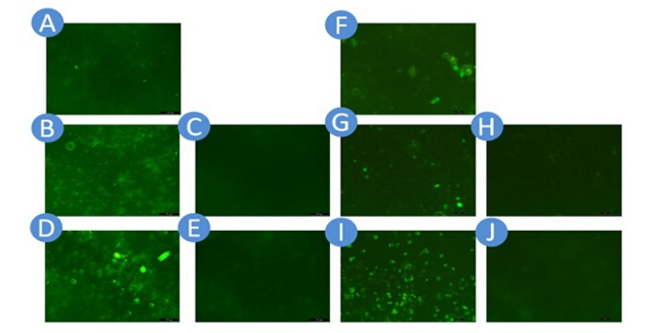
Fig. 12: Green flurescent light produced by GFP reporter gene.
A-E are T24 cell line and F-J are 5637 cell line. A&F: Positive Control Group B&G: Two Plasmids System with Ack C&H: Two Plasmids System without Ack D&I: Three Plasmids System with Ack E&J: Three Plasmids System
Discussion
Our team had been working to find a kind of effective and without side effects or fewer side effects gene therapy method to the directional kill bladder cancer cell precisely since Feb. 2015. Through a large number of literature theory support, we combined AND gate with unnatural amino acids(UAA) orthogonal system and constructed a gene circuit, which has two switches, namely the cancer cell specific promoter hTERT and the bladder cancer cell specific promoter hUPll. Only they are started simultaneously can drive the downstream gene expression.
We constructed two plasmids and three plasmids expression system and tested with different bladder cancer cell lines and human normal epithelial cells(HFC) and found that, the expression ability was no different in the two plasmids and the three plasmids expression system. No expression in HFC indicated that the genetic circuit system has no effect to HFC.
As we all know, gene therapy strategies have been developed, we hope to develop a precise and effective and without side effects gene circuit method into the bladder cancer. We are convinced that gene therapy will be our first choice for many diseases treatment in the near future.
Conclusion
These findings suggest that the constructed circuit based on AND GATE and UAA orthogonal system can be used to specifically identify bladder cancer cells and express therapeutic gene only when Ack exist. These results may yield a new therapeutic approach for bladder cancer.
Acknowledgments
The authors would like to thank all members of 2015 SZU-iGEM team from China for their constructive efforts, and thank the team instructors and advisors for their criticisms and support.
Financial Disclosure
The Second People’s Hospital of Shenzhen supported our work in designing the study and providing laboratory apparatus.
This work was also supported by Educational Administration Centre, International Office, Alumni Association and Student Affairs Office of Shenzhen University, GENZON, Junneng Industrial Co., Ltd. and Sunwelcome Technology. These funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing Interests
The authors have declared that no competing interests exist.
Data Availability
Yes – all data are fully available without restriction
References
[1] Falke J, Witjes JA. Contemporary management of low-risk bladder cancer. Nat Rev Urol. 2011;8:42–9.
[2] Lei AQ, Cheng L, Pan CX. Current treatment of metastatic bladder cancer and future directions. Expert RevAnticancer Ther. 2011;11(12):1851–62.
[3] Marta GN, Hanna SA, Gadia R, Correa SF, Silva JL, Carvalho Hde A. The role of radiotherapy in urinary bladder cancer: current status. Int Braz J Urol. 2012;38(2):144–53.
[4] https://en.wikipedia.org/wiki/AND_gate
[5] Mano,M.Morris and Charles R.Kime. Logic and Computer Design Fundamentals, Third Edition.Prentice Hall, (2004)
[6] Yuchen Liu,Yayue Zeng,Li Liu,Chengle Zhuang,Xing Fu,Weiren Huang& Zhiming Cai.Synthesizing AND gate genetic circuigts based on CRISPR-Cas9 for identification of bladder cancer cells. DOI:10.1038/ncomms6393(2014)
[7] Yoshida,W.&Yokobayashi,Y.Photonic Boolean logic gates based on DNA aptamers. Chem.Commun.(Camb.)14,195–197(2007)
[8] Wang,B.,Kitney,R.I.,Joly,N.&Buck,M.Engineering modular and orthogonal genetic logic gates for robust digital-like synthetic biology. Nat.Commun. 2, 508 (2011).
[9] Mcelroy D, Brettell R. Foreign gene expression in transgenic cereals[J]. Trends, Biotechnol, 1994,12(2):62-68
[10] Jongchan W, Matthew HH, Albrecht G. Structure-function studies on the active site of the coelenterazine-dependent luciferase from Renilla, Proteinscience, 17(10): 725-735
[11] P.A.V. Anderson, J.F. Case, Electrical activity associated with luminescence and other colonial behaviour in the pennatulid Renilla kollikeri, Biol. Bull. 149(1975): 80–95.
[12] Chang C. Liu and Peter G. Schultz,Adding new chemistries to the genetic code,The Scripps Research Institute, La Jolla, California 92037
[13]GE Yongbin, HONG Jiong, WANG Dongmei Orthogonal genetic system in synthetic biology Department of Physics and Chemistry, Bozhou Normal College, Bozhou 236800, China;School of Life Science, University of Science and Technology of China, Hefei 230026, China
[14]Appella DH. Non-natural nucleic acids for synthetic bi-ology. Curr Opin Chem Biol, 2009, 13: 687-696
[15]An W, Chin JW. Synthesis of orthogonal transcrip-tion-translation networks. Proc Natl Acad Sci USA, 2009, 106: 8477-8482
[16] Nivedita Mitra ,Incorporating Unnatural Amino Acids into Recombinant Proteins in Living Cells. Yale University, United States, MATER METHODS 2013;3:204
[17] Li, Jie Wang, Jie Chen, Peng R.* Unnatural Amino Acid Mediated Protein Bioorthogonal Labeling. ACTA CHIMICA SINICA 70, 1439—1445(2012)